AMROCE will develop antimicrobial/antibiofilm fish cage nets and wastewater filtration membranes through polymer bulk and surface nano-engineering. Marine-derived antimicrobial agents and antibiofilm enzymes will be nano-formulated as alternative to antibiotics for fish and animal feed supplement. Human and environmental nanosafety during the manufacturing and use of the novel nanotechnology-embedded products will be continuously evaluated to anticipate nanosafety issues.
The Main Objective of the AMROCE project is to develop novel antibiotic-free antimicrobial strategies to improve the animal and fish living conditions, reducing the use of antibiotics, the accumulation of antibiotics in the environment and the emergence of antimicrobial resistant (AMR). These strategies comprise:
- bulk engineering of antimicrobial and antibiofilm fish cage nets (FCN) and water filtration membranes (WFM) from thermoplastic polymers blended with antibacterial to Metal Oxide Nanoparticles (MeO NPs),
- surface nano-engineering of FCN and WFM using NP-based antimicrobial and antibiofilm coatings,
- nano-formulation of antibiofilm enzymes and marine-derived antimicrobial lipids and AMP for fish and animal feed.
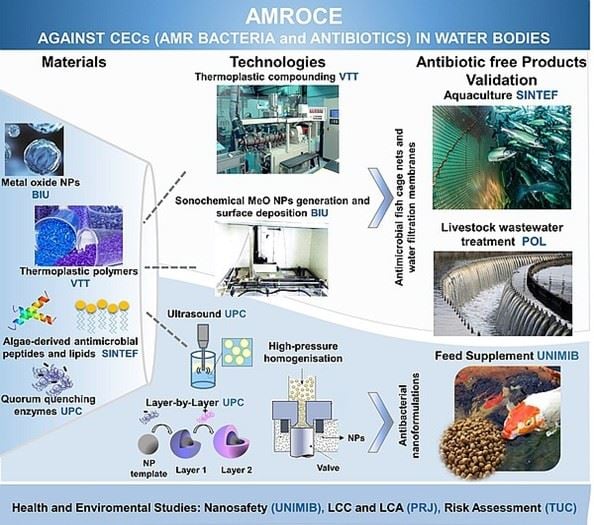
Figure 1. AMROCE layout.
Activities in AMROCE project is divided in 7 work packages (WP):
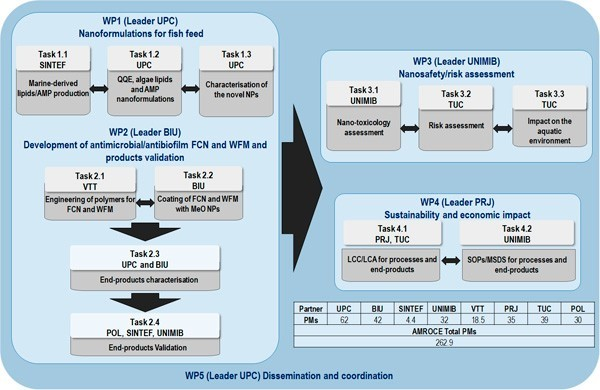
Figure 2. AMROCE workflow, partners’ interactions and complementarity.
Activities with Norwegian partners involved
SINTEF Ocean is responsible for task 1.1. SINTEF will screen for novel lipids and AMP (antimicrobial proteins) as an antimicrobial bio-based platform for nanoformulation of QQE (quorum quenching enzymes). Selected algae e.g. Dunaliella, Chlorella or Haematococcus will be cultivated in small-scale bioreactors (< 1 L) under different stress conditions to induce the production of antimicrobial secondary metabolites. Processing techniques, including protease-assisted freeze-drying, bed-milling, US and pressurised extraction, will be optimised to improve the extraction efficacy and the quality of the secondary metabolites. Purification by chromatographic methods (e.g. HPLC) will yield peptide and lipid samples further analysed and quantified by nuclear magnetic resonance (NMR), liquid chromatography, mass spectrometry (MS), gas chromatography (GC)-FID, and GC-MS/MS among others. Partner UPC will screen the isolated lipids and peptides for antimicrobial and antibiofilm activity.
As a part of task 2.4 NTNU will work to verify WFM and FCN developed in tasks 2.1 and 2.2 antifouling effect in sea-based aquaculture, small sections of coated (test) and uncoated (control) net pens will be placed in close proximity to the salmon aquaculture farm NTNU uses for their R&D license M-VS-00016. Sample nets will be distributed randomly in mounting frames. Biofilm formation and fouling development will be measured repeatedly weekly to monthly, with short sampling intervals in the beginning to monitor biofilm formation and longer time intervals later in the tests to follow a succession of larger fouling organisms. Biofilm will be studied by microscopy to determine the ratio of prokaryotes and eukaryotes, and NGS metagenomics analysis to study the composition of the prokaryote microbiome and the difference between treated and untreated nets. Potential macro-fouling will be evaluated by measuring the percent of net aperture occlusion and by determining the fouling species and their contribution to the total amount of fouling.
Project funding: The project is funded by EU and Norwegian Research Council for funding, in the frame of the collaborative international consortium AMROCE financed under the ERA-NET AquaticPollutants Joint Transnational Call (GA Nº 869178). This ERA-NET is an integral part of the activities developed by the Water, Oceans and AMR Joint Programming Initiatives.







