Renewable energy sources and increase of energy efficiency have been named as the most important instrument to solve the global climate challenges and secure energy supply. Due to the intermittent nature of wind and solar power, new technologies for large scale energy storage and grid balancing will become essential to allow the renewable energy input in Europe to grow to desired levels.
Liquid metal batteries (LMB) have great potential for high throughput (power density) due to the all liquid system. It is not a new concept for energy storage, but gained worldwide attention in 2009, when the US Depart. of Energy's new Advanced Research Projects Agency (ARPA-E) announced a massive economic support to this type of battery research to the group of Professor D. Sadoway at Massachusetts Institute of Technology (MIT). Current research in the field of liquid metal batteries focuses on batteries with no membrane or diaphragm using metal/metal-alloy systems which results in a severe voltage penalty. Another type of LMB is the State-of-the-art sodium sulphur (NaS) molten salt batteries which have been demonstrated applicable for grid storage. They require a sodium ion selective membrane which adds a great deal of expense and resistance to the cell. In addition, in the event of rupture or cracking of the membrane a vigorous reaction occurs, possibly resulting in fires.
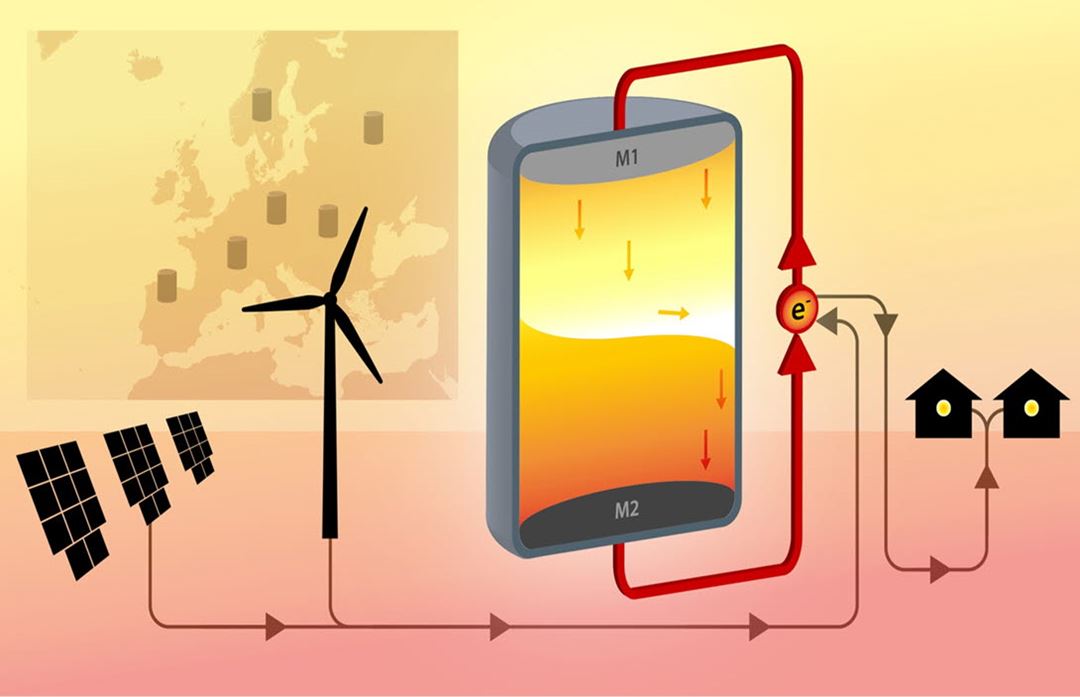
In our project, we introduce a novel concept of producing a "membrane-free" sodium-zinc based all liquid battery. The battery consists of three liquid layers; sodium at the top as the negative electrode, a sodium chloride based electrolyte in the middle, and zinc at the bottom as the positive electrode. To prevent the zinc containing ions from reacting with the sodium electrode, a porous diaphragm or separator is placed between the electrodes. The exclusion of the brittle, expensive β-alumina ion selective membrane and its replacement with a cheap durable diaphragm material will significantly improve both performance and reduce the cost level of liquid metal batteries. The choice of immiscible electrolytes and electrodes will ensure a safe battery system, which in the unlikely event of mechanical failure will discharge without any undesired effects such as fire or explosion.
SINTEF works partly close with NTNU on this project on lab scale cell design development, testing of battery performance and materials. Our broad experience on molten salt electrochemistry and electrolysis is essential to obtain this.
Partner: NTNU, Department of Materials Science and Engineering.
Contact at NTNU, Professor Geir Martin Haarberg
The project is funded by The Research Council of Norway